The physics behind the most ordinary miracle in the sky — and how that sky has changed, in color, across 4.5 billion years of Earth history
The Question Everyone Has Stopped Asking
You know the sky is blue. You’ve known it your whole life. And because you’ve known it your whole life, you’ve probably stopped asking why. That’s a shame, because the answer is one of the most elegant pieces of physics in nature — and once you understand it, you start seeing the sky differently. Not just today’s sky, but the sky as it has been across geological time: orange and brown and hazy and alien, and only very recently the familiar blue that feels, to us, like the obvious color a sky should be.
It was never obvious. For most of Earth’s history, the sky was not blue. The shift to blue was one of the most dramatic transitions in the history of a planet, driven by microscopic organisms that didn’t set out to repaint the atmosphere. They just wanted to eat sunlight.
But first: the physics.
Rayleigh Scattering: The Mechanism
Sunlight is not a single color. It is a mixture of all visible wavelengths — from violet (roughly 380 nanometers) through blue, green, yellow, and orange to red (roughly 700 nm), plus wavelengths beyond either end that human eyes cannot see. When that light enters Earth’s atmosphere, it encounters the molecules of nitrogen (N₂) and oxygen (O₂) that make up roughly 99% of the air. Those molecules are tiny — about a tenth of a nanometer across — hundreds of times smaller than even the shortest wavelength of visible light. When an electromagnetic wave encounters a particle far smaller than its wavelength, the oscillating electric field of the wave sets the charges within that particle vibrating at the same frequency. The particle becomes a tiny radiating dipole, re-emitting the light in all directions. This is Rayleigh scattering, named for the British physicist John William Strutt, Lord Rayleigh, who worked out the mathematics in 1871 and 1881 and confirmed its molecular basis in 1899.
The critical insight is the wavelength dependence: the intensity of Rayleigh scattering is inversely proportional to the fourth power of the wavelength. Not the second power. The fourth power. That is an enormous sensitivity. Red light, with a wavelength roughly twice that of blue light, is scattered about 16 times less efficiently. Blue light at around 450–480 nm is flung sideways across the entire sky. Red and orange light mostly passes straight through the atmosphere to your eyes.
That’s why the sky appears blue: you’re looking at scattered light, not direct sunlight. In every direction away from the sun, you are seeing the blue end of the spectrum that has been re-radiated sideways by billions of air molecules throughout the atmospheric column above you.
| Expert Capsule — Why Not Violet? The Eye, the Sun, and the Ozone Layer Violet light (wavelength ~380–420 nm) is scattered even more efficiently than blue by Rayleigh scattering, since it has a shorter wavelength. So why does the sky appear blue rather than violet? Three factors work together. First, the Sun emits less violet than blue — its emission spectrum peaks in the blue-green range, so more blue light enters the atmosphere to begin with. Second, the Earth’s ozone layer and upper atmosphere absorb a significant fraction of incoming violet and ultraviolet wavelengths before they can scatter at lower altitudes, further depleting violet contribution to the daytime sky. Third, human color vision uses three types of cone cells; the short-wavelength cone is not exclusively violet-sensitive but responds across a range including blue, and neural color processing combines signals from all three cone types. The resulting perception of the scattered mixture — depleted violet, abundant blue — is experienced as blue. Our visual system is a color-comparison machine, not a spectrometer. |
Sunsets and the Long Way Round
Sunsets are the same physics, viewed at an extreme angle. When the sun is near the horizon, its light must travel through a far thicker column of atmosphere to reach your eyes — the path length grows to tens of times the noon value. By the time it arrives, nearly all the blue has been scattered out along the way, leaving the longer wavelengths — red, orange, and yellow — to dominate. The sun turns red, and the low sky does too.
Large volcanic eruptions can intensify sunsets dramatically for months or years, by loading the stratosphere with sulfate aerosol particles. These particles are much larger than air molecules and scatter light by Mie scattering rather than Rayleigh scattering — a less wavelength-selective process, but one that enhances extinction of shorter wavelengths and produces extraordinary reds. The 1815 eruption of Mount Tambora in Indonesia is widely cited as responsible for the vivid red skies recorded in J. M. W. Turner’s paintings in the years that followed.
| Expert Capsule — Rayleigh vs. Mie Scattering: Why Particle Size Is Everything The distinction between Rayleigh and Mie scattering governs sky color across the solar system. Rayleigh scattering applies when the particle is much smaller than the wavelength of light (~1/10 or less): scattering intensity scales as λ⁻⁴, giving strongly wavelength-dependent, blue-biased sky color. Mie scattering applies when particle size approaches the wavelength of light — as with dust grains, fog droplets, volcanic aerosols, and the iron-oxide particles of the Martian atmosphere. Mie scattering is far less wavelength-selective: it distributes visible wavelengths more uniformly, with the absorption properties of the particles, rather than scattering physics alone, determining the sky color. Hadean sulfate aerosols, Martian iron-oxide dust, and Titan’s tholin haze all represent Mie or near-Mie regimes. Earth’s blue sky is specifically the result of a Rayleigh-dominated atmosphere — one cleared of large aerosols and composed primarily of small gas molecules. |
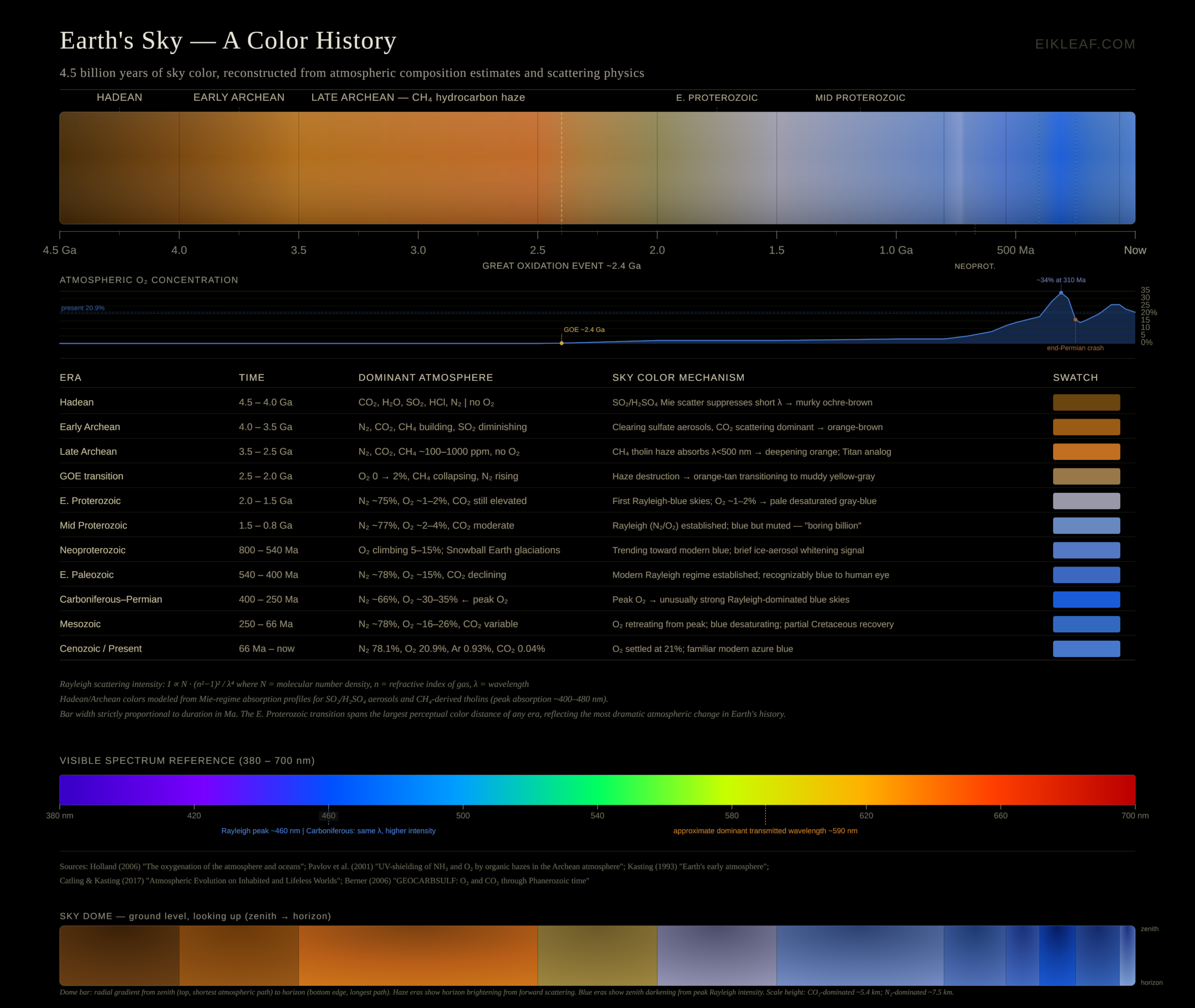
The Sky’s Color History: 4.5 Billion Years
The sky has not always been blue. In fact, for most of Earth’s history it was not blue at all, and the progression from the early Hadean to today is one of the most dramatic stories in planetary science. The infographic accompanying this article charts that progression. The colors assigned to each era below are modeled reconstructions — inferred from atmospheric composition estimates, scattering physics, and analogs like Titan — not direct observations. With that caveat stated, the broad picture is well-grounded in the literature.
The Hadean (4.5 – 4.0 Ga): Ochre-Brown and Murky
When Earth formed 4.5 billion years ago, it had no sky in any recognizable sense. The surface was largely molten, bombarded by debris from the early solar system. As the planet cooled and volcanic outgassing began building an atmosphere, the dominant gases were CO₂, water vapor, sulfur dioxide (SO₂), hydrogen chloride (HCl), and nitrogen — with essentially no free oxygen, present at only about 0.001% of today’s atmospheric level.
The sulfur dioxide was almost certainly crucial to the sky’s modeled color. SO₂ reacts with water vapor to form sulfuric acid (H₂SO₄) aerosol particles — the same chemistry that gives Venus its characteristic cloud cover today. These aerosol particles are much larger than air molecules, placing scattering in the Mie rather than Rayleigh regime. The modeled result: a murky ochre or brown sky, optically thick, sulfurous, and essentially impenetrable.
The Early Archean (4.0 – 3.5 Ga): Fading Brown
As volcanic activity gradually declined and the planet stabilized, sulfur dioxide levels fell. Sulfate aerosol concentrations diminished and the heavy Mie-scattering haze began to thin. The sky’s composition shifted toward CO₂ and N₂ as dominant gases. Enough residual aerosol likely remained to mute any nascent blue signal. The modeled sky during this era is a warm, hazy orange-brown — cleared from the deep murk of the Hadean, but still far from blue.
The Late Archean (3.5 – 2.5 Ga): Orange — A Living Haze
Life appears in the geological record by at least 3.5 billion years ago. The earliest organisms were anaerobic, and many were methanogens: microbes that produced methane (CH₄) as a metabolic byproduct. As these organisms spread, methane likely accumulated in the atmosphere at concentrations estimated between 100 and 1,000 parts per million. Ultraviolet light from the Sun photodissociates methane molecules, and the resulting reactive fragments polymerize into complex organic aerosol particles called tholins: the same reddish-brown, tar-like haze that gives Saturn’s moon Titan its characteristic color today.
This tholin haze efficiently absorbs short-wavelength light below roughly 500 nm, stripping blue and violet from the sky and leaving only longer, warmer wavelengths. The modeled result is a sky of deepening orange. The Titan analogy is not merely rhetorical: Titan’s nitrogen-methane atmosphere with abundant tholins is the closest existing analog to what the Late Archean sky likely looked like. Modern coupled climate-photochemical-microphysical simulations confirm that hydrocarbon haze of this kind is consistent with habitable surface conditions despite the fainter young Sun, which itself shone at only about 70% of its current luminosity at this time.
| Expert Capsule — The Faint Young Sun Paradox and the Methane Greenhouse The Sun was roughly 70–80% as luminous 4 billion years ago as it is today — a well-established consequence of stellar evolution. By straightforward radiative balance, this should have left early Earth well below freezing. Yet geological evidence is unambiguous that liquid surface water existed throughout the Archean. The most plausible resolution is a powerful greenhouse effect from CO₂ and, critically, from the high methane levels produced by methanogens. A dense methane-tholin haze would simultaneously have created the orange sky modeled for this period and helped maintain surface temperatures above freezing — a remarkable case of life inadvertently engineering its own climate. When the Great Oxidation Event destroyed the methane greenhouse, Earth experienced its first major glaciation, the Huronian, roughly 2.3–2.1 billion years ago. |
The Great Oxidation Event (~2.4 Ga): The Hinge of History
Around 2.46 to 2.43 billion years ago, Earth crossed the most consequential atmospheric threshold in its history: the Great Oxidation Event (GOE). Free oxygen began accumulating permanently in the atmosphere for the first time, produced by cyanobacteria — the first organisms capable of oxygenic photosynthesis, splitting water molecules and releasing O₂ as a byproduct.
The effect on the sky was transformative. Oxygen reacted with the abundant atmospheric methane, destroying the tholin haze chemistry almost entirely. The orange sky — which had persisted for over a billion years — dissolved. But what replaced it was not blue, not immediately. With oxygen still at just 1–2% of the atmosphere (compared to today’s 20.9%), the sky entered a transitional phase: murky yellow-gray to tan, progressively clearing as the aerosol load dropped. The methane collapse also triggered the Huronian glaciation — a global ice age caused by the destruction of the methane greenhouse, compounded by the faint young Sun.
The scale of this event is difficult to overstate. Microscopic single-celled organisms, pursuing nothing more than the chemistry of extracting energy from sunlight and water, changed the color of the entire sky. They changed the chemistry of the oceans. They triggered a global ice age. They drove most of the then-existing anaerobic life to extinction. And they laid the foundation for every complex multicellular organism that would ever exist — including the ones now reading these words.
The Early Proterozoic (2.0 – 1.5 Ga): The First Blue Sky
As oxygen stabilized at around 1–2% of the atmosphere, the composition shifted toward something increasingly familiar: roughly 75% nitrogen, with oxygen as a meaningful minority component. For the first time, Rayleigh scattering by N₂ and O₂ molecules could dominate the sky. The sky became blue — but a pale, desaturated gray-blue, not today’s deep azure. At 1–2% oxygen and with CO₂ still elevated, the Rayleigh signal was weaker than modern. Still: this was, as far as we can reconstruct, the first genuinely blue sky on Earth.
The “Boring Billion” (1.5 – 0.8 Ga): Steady Blue
The period from about 1.5 to 0.8 billion years ago is sometimes called the ‘boring billion’ by geologists — an epoch of remarkable atmospheric stability, with oxygen holding at roughly 2–4% and biological innovation proceeding slowly. The sky was modeled to be blue but muted, lacking the vivid saturation of today’s atmosphere. A human observer transported to this era would recognize the sky as blue, but pallid — a washed-out version of the familiar.
The Neoproterozoic and Cambrian (800 – 540 Ma): Approaching Modern
Oxygen rose progressively through the Neoproterozoic, from roughly 5% toward 15%, as complex multicellular life proliferated and oceanic productivity expanded. The Snowball Earth glaciations of this period (roughly 720–635 Ma) likely created transient sky anomalies: ice-aerosol whitening during glacial maxima. By the onset of the Cambrian explosion around 540 million years ago, oxygen had reached approximately 15% and the modeled sky would have been recognizably similar to today’s — blue, clear, with the familiar zenith-to-horizon gradient.
| Expert Capsule — How Oxygen Concentration Affects Sky Color The intensity of Rayleigh scattering depends on the number density of scattering molecules and their polarizability — the ease with which electron clouds are displaced by an electromagnetic field. O₂ has a somewhat higher polarizability than N₂, so an atmosphere richer in oxygen scatters blue light more intensely for a given total pressure. At the Carboniferous peak of ~30–35% O₂ (versus today’s 20.9%), the sky would likely have been a somewhat more saturated blue — not dramatically different to the naked eye, but measurably deeper. This is a directional claim well-grounded in scattering physics; the exact visual difference is modeled, not measured. |
The Carboniferous and Permian (400 – 250 Ma): Likely the Deepest Blue
The most intensely blue sky in Earth’s post-Hadean history was likely during the Carboniferous and Permian periods, roughly 310–260 million years ago. The evolution of large woody vascular land plants — the coal-forming forests of the Carboniferous — buried vast quantities of organic carbon, drawing down CO₂ and simultaneously producing elevated atmospheric oxygen. Robert Berner’s GEOCARBSULF model, the most widely cited quantitative reconstruction of Phanerozoic atmospheric composition, calculates oxygen reaching approximately 30–35% during this interval — the highest modeled level in the Phanerozoic record. The coal seams that powered the Industrial Revolution are the compressed, fossilized remains of those forests.
A roughly 67% increase in oxygen concentration above today’s level, combined with the higher polarizability of O₂ relative to N₂, would have produced measurably stronger Rayleigh scattering and a more saturated blue sky than today’s. The same oxygen levels contributed to the legendary giant arthropods of the period: Meganeura dragonflies with 70-centimeter wingspans, and Arthropleura millipedes over two meters long, whose respiratory systems could support greater body mass in a hyperoxic atmosphere. The Permian mass extinction 252 million years ago — the most severe in Earth’s history, eliminating an estimated 96% of marine species — coincided with a sharp drop in atmospheric oxygen. Whatever blue the coal swamps had overhead, it did not persist.
The Mesozoic and Cenozoic (250 Ma – Now): Settling to Azure
After the Permian extinction, oxygen recovered gradually through the Mesozoic — the age of dinosaurs. It oscillated between roughly 16% and 26% through the Triassic, Jurassic, and Cretaceous, never returning to Carboniferous levels. The sky varied in saturation accordingly, but was always recognizably blue. By 66 million years ago, as the Chicxulub impact ended the Cretaceous, oxygen was settling toward the modern value. The Cenozoic saw it stabilize near 20.9%, producing the familiar deep azure of a clear midlatitude day.
Today’s atmosphere is 78.1% nitrogen, 20.9% oxygen, 0.93% argon, and 0.04% CO₂. The sky color we regard as self-evidently normal is, in geological terms, an extremely recent achievement. For roughly the first 2 billion years of Earth’s history, there was no blue sky. For another billion, the blue was pale and uncertain. The vivid blue of a clear summer day — the blue of high altitude, of the cover of every science magazine — is a gift from cyanobacteria, delivered across geological time.

Other Skies: What the Universe Offers
Earth’s blue sky is unusual, not inevitable. The variety of sky colors on the planets and moons we have visited — or can constrain from atmospheric data — shows that sky color is a specific, contingent outcome of atmospheric composition, particle size distribution, and stellar spectrum. Here are the most instructive cases, all grounded in direct measurement or tightly constrained models.
Mars: Butterscotch Days, Blue Sunsets
Mars is the most instructively inverted case. The Martian atmosphere is approximately 95% CO₂, but surface pressure is only about 0.6% of Earth’s, making Rayleigh scattering from gas molecules negligible. Instead, the sky is shaped by fine iron-oxide-rich dust particles — roughly 1–3 microns in diameter, about the consistency of talcum powder — suspended permanently by weak Martian gravity (38% of Earth’s) and replenished continuously by dust storms. These particles absorb blue and green light efficiently while scattering longer wavelengths more uniformly, producing the yellow-brown to pinkish-orange sky that Mars rovers have consistently measured — a color NASA scientists have described as ‘butterscotch.’
At sunset the geometry reverses. The forward-scattering character of Mie scattering by the dust particles concentrates blue-scattered light near the Sun’s disk, producing a distinctive blue glow surrounded by the reddish sky. Mars has blue sunsets and orange days: the precise opposite of Earth. This inversion has been directly imaged by the Opportunity, Spirit, and Curiosity rovers. If all Martian dust were somehow removed, the sky would revert to a very dark, nearly black blue — the Rayleigh signal from the thin CO₂ gas alone, barely enough to register.
| Expert Capsule — If Mars Had No Dust Planetary scientists have modeled what the Martian sky would look like if the atmosphere were cleared of all dust. The answer is striking: the sky would be very dark blue — darker than Earth’s high-altitude sky — because the Martian atmosphere is so thin that far fewer scattering events occur per unit path. From the surface, you could likely see bright stars in the daytime. The horizon would be almost black. The Sun would appear as a sharp, slightly yellowish disk. Mars’s dust is not just a nuisance: it is the entire mechanism by which the planet has a visible sky color at all. |
Titan: The Living Archive of Earth’s Archean Sky
Saturn’s moon Titan is the most direct observational analog to Earth’s Late Archean atmosphere available in the solar system. Its atmosphere is approximately 98.4% nitrogen and 1.4% methane — structurally analogous to what Earth’s atmosphere is reconstructed to have been before the GOE, though much colder and receiving far less solar energy. UV photolysis of methane in Titan’s upper atmosphere continuously produces tholin haze particles: the same class of complex organic molecules modeled for Archean Earth.
The result, measured directly by the Huygens probe’s Descent Imager/Spectral Radiometer during its 2005 descent through Titan’s atmosphere, is a sky dominated by Mie scattering from the multilayer tholin haze — aggregate fractal particles with monomer radii of roughly 0.05 microns, distributed from the surface to over 150 km altitude. The sky is approximately 100–1,000 times dimmer than an Earth afternoon and uniformly dark orange in all directions. Sunsets on Titan are described by atmospheric models as ‘underwhelming’ — the Sun simply fades into the haze without distinct color change. The Dragonfly rotorcraft mission, planned to reach Titan around 2034, will study this chemistry directly.
Venus: Dim Orange-Yellow Under Sulfuric Clouds
Venus has an atmosphere roughly 90 times denser than Earth’s, composed almost entirely of CO₂, with thick clouds of sulfuric acid droplets (H₂SO₄) at 45–70 km altitude. The surface receives only about 2–3% of the sunlight that reaches Earth’s surface. Lander images from the Soviet Venera probes show a sky that appears dim orange to yellowish-brown at the surface, filtered through H₂SO₄ clouds that absorb blue and violet light and scatter the remaining longer wavelengths downward. The surface itself, illuminated by this filtered light, appears in the Venera images as orange-red rock under a perpetually warm, hazy sky. In structure, Venus’s atmosphere today is a compressed and intensely hot version of the Hadean Earth: dominated by CO₂ and sulfur chemistry, with sky color governed by sulfate aerosols rather than gas-phase Rayleigh scattering.
Exoplanets: A Full Palette
Beyond the solar system, sky color is contingent on so many variables that almost any color is physically plausible. The host star’s emission spectrum is a primary driver: a planet orbiting a cool M-dwarf star — which emits mostly red and infrared light, with relatively little blue — would produce a more pinkish or lavender sky even with an Earth-composition nitrogen-oxygen atmosphere, simply because there is less blue starlight to scatter in the first place. Atmospheric pressure matters too: a very high-pressure atmosphere produces denser scattering and a brighter, more saturated sky; a thin one produces a dark, near-black sky.
HD 189733b, a hot Jupiter at roughly 63 light-years, appears deep blue in reflected albedo measurements — but not from Rayleigh gas scattering. Its blue comes from silicate cloud particles (effectively tiny glass spheres) high in the atmosphere, which scatter blue light strongly. The dayside temperature approaches 1,200°C and winds exceed 8,000 km/h. It is blue without being anything like a livable sky. The point is that sky color is a readout of atmospheric physics, not an indicator of habitability.
| Expert Capsule — What Would a Green Sky Require? Green light (~520–560 nm) sits in the middle of the visible spectrum: scattered less efficiently than blue by Rayleigh scattering, but more so than red. A sky dominated by green is essentially impossible through Rayleigh scattering alone — any atmosphere that preferentially scatters green would scatter blue and violet even more strongly, and those shorter wavelengths would dominate. A genuinely green sky would require an absorber that removes blue from the sky while transmitting green — acting as a spectral filter. Some researchers have noted that before the Great Oxidation Event, widespread marine photosynthesis could have produced enough chlorophyll-bearing aerosol particles to create a very subtle green tinge, but this remains speculative. A truly saturated green sky is a chemistry problem, not a physics problem: you need the right absorber, not just the right scatterer. |
The Blue We Inherited
The blue sky is not a given. It is not what skies look like — it is what this particular sky looks like, at this particular moment in geological time, after a specific and improbable chain of events: the origin of oxygenic photosynthesis, the Great Oxidation Event, the progressive rise of an oxygen-nitrogen atmosphere to its current composition, and the stabilization of that composition over the past hundred million years.
Every time you look up at a clear sky and see blue, you are seeing the accumulated byproduct of 2.4 billion years of cyanobacterial metabolism. You are looking at the chemical signature of life’s most consequential invention. The sky is not merely a backdrop to the story of life on Earth. The sky is part of that story. It was made by life, changed by life, and it remains today a direct readout of the living chemistry of this planet.
Lord Rayleigh worked out the physics in 1871. The cyanobacteria did the engineering starting around 2.7 billion years ago. And we get to stand underneath the result and say: yes, that color — that’s what a sky looks like.
Gen AI Disclaimer
Some contents of this page were generated and/or edited with the help of a Generative AI.
Media
Ten Clear to Cloudy Hot Jupiters – NASA
Key Sources and References
Bohren, C. F., & Huffman, D. R. (1983). Absorption and Scattering of Light by Small Particles. Wiley-Interscience. ISBN 0471057727. (Rayleigh and Mie scattering theory; wavelength-dependence of scattering; particle-size regimes; sulfate aerosol scattering; all scattering physics throughout the article.)
Chandrasekhar, S. (1960). Radiative Transfer. Dover Publications. ISBN 0486605906. (Classical mathematical treatment of radiative transfer and Rayleigh scattering in planetary atmospheres.)
Young, A. T. (1982). Rayleigh scattering. Physics Today, 35(1), 42–48. doi:10.1063/1.2890003. (Authoritative treatment of Rayleigh scattering terminology, history, and the molecular basis established by Rayleigh in 1899; clarifies elastic vs. inelastic scattering distinctions.)
Nassau, K. (1983). The Physics and Chemistry of Color: The Fifteen Causes of Color. Wiley-Interscience. ISBN 0471867764. (Sky color and color perception; blue-vs.-violet discussion; human cone sensitivity and neural color processing.)
Pavlov, A. A., Kasting, J. F., Brown, L. L., Rages, K. A., & Freedman, R. (2001). UV shielding of NH₃ and O₂ by organic hazes in the Archean atmosphere. Journal of Geophysical Research, 106(E10), 23267–23287. (Methane-derived hydrocarbon haze in the Archean; optical properties and UV-shielding role; basis for the orange Late Archean sky reconstruction.)
Arney, G., Domagal-Goldman, S. D., Meadows, V. S., Wolf, E. T., Schwieterman, E., Charnay, B., Claire, M., Hebrard, E., & Trainer, M. G. (2016). The Pale Orange Dot: The Spectrum and Habitability of Hazy Archean Earth. Astrobiology, 16(11), 873–899. doi:10.1089/ast.2015.1422. (Coupled climate-photochemical-microphysical simulations of hazy Archean Earth; confirmed orange-sky model and habitable surface conditions; Titan analog; faint young Sun context.)
Sessions, A. L., Doughty, D. M., Welander, P. V., Summons, R. E., & Newman, D. K. (2009). The continuing puzzle of the Great Oxidation Event. Current Biology, 19(14), R567–R574. doi:10.1016/j.cub.2009.08.002. (GOE timing and mechanisms; O₂ < 10⁻⁵ PAL pre-GOE, rise to 1–10% PAL during the event; atmospheric transition narrative.)
Lyons, T. W., Reinhard, C. T., & Planavsky, N. J. (2014). The rise of oxygen in Earth’s early ocean and atmosphere. Nature, 506, 307–315. doi:10.1038/nature13068. (Gold-standard review of Earth’s oxygenation history; Proterozoic and Neoproterozoic atmospheric constraints; Huronian glaciation context.)
Berner, R. A. (2006). GEOCARBSULF: A combined model for Phanerozoic atmospheric O₂ and CO₂. Geochimica et Cosmochimica Acta, 70(23), 5653–5664. doi:10.1016/j.gca.2005.11.032. (Quantitative reconstruction of Phanerozoic O₂; Carboniferous peak ~30–35% O₂; Permian–Triassic crash; basis for Carboniferous sky saturation discussion.)
Beerling, D. J., & Berner, R. A. (2000). Impact of a Permo-Carboniferous high O₂ event on the terrestrial carbon cycle. PNAS, 97(23), 12428–12432. doi:10.1073/pnas.220280097. (~35% O₂ during Permo-Carboniferous confirmed; giant insect physiology and wildfire ecology at elevated oxygen.)
Tomasko, M. G., Doose, L., Engel, S., Dafoe, L. E., West, R., Lemmon, M., Karkoschka, E., & See, C. (2008). A model of Titan’s aerosols based on measurements made inside the atmosphere. Planetary and Space Science, 56(5), 669–707. doi:10.1016/j.pss.2007.03.006. (Direct measurement of Titan’s haze from the Huygens probe DISR instrument; aggregate fractal particles, monomer radius ~0.05 microns; sky brightness 100–1000x below Earth; basis for Titan sky description.)
Titov, D. V., Ignatiev, N. I., McGouldrick, K., Wilquet, V., & Wilson, C. F. (2018). Clouds and hazes of Venus. Space Science Reviews, 214, 126. doi:10.1007/s11214-018-0552-z. (Venus cloud structure and optical properties; H₂SO₄ cloud absorption of blue-violet wavelengths; basis for Venera surface sky description.)
Pierrehumbert, R. T. (2010). Principles of Planetary Climate. Cambridge University Press. (Atmospheric physics of planetary climates; Hadean and Archean atmospheric modeling context; faint young Sun and greenhouse gas discussion.)
Seager, S. (2010). Exoplanet Atmospheres: Physical Processes. Princeton University Press. (Exoplanet atmospheric physics; M-dwarf star sky color discussion; HD 189733b silicate cloud scattering context.)
Ulfur Atli
Writing mainly on the topics of science, defense and technology.
Space technologies are my primary interest.






